CBS News’ 60 Minutes recently featured the Rockefeller Neuroscience Institute’s groundbreaking work and research.
Tomorrow’s breakthroughs in brain health will happen here at the WVU Rockefeller Neuroscience Institute.
RNI DEPARTMENTS
Neuroradiology
Learn more →
Behavioral Medicine & Psychiatry
Learn more →
Children’s Neuroscience Center
Learn more →
SPECIALIZED NEUROLOGICAL CARE
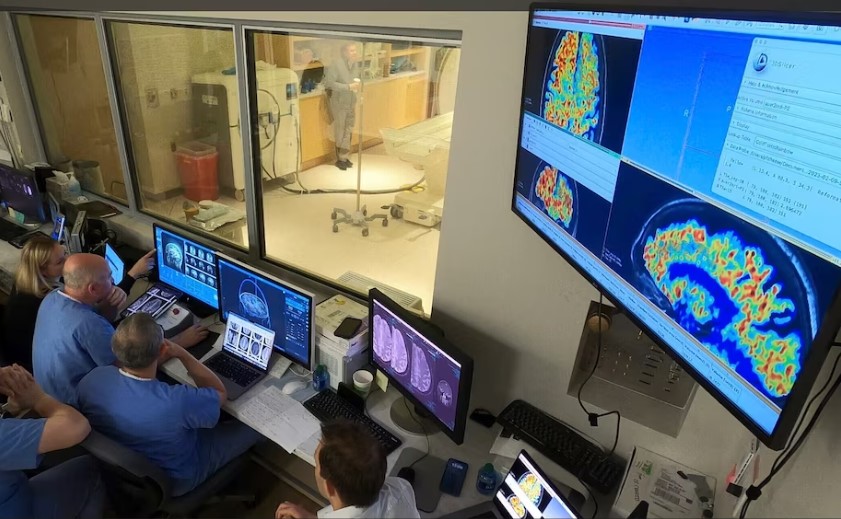
RNI Research
The RNI Innovation Center research team is a dedicated multidisciplinary group of clinicians, scientists, engineers, clinical trial specialists, and research staff focused on translational clinical trials. This team partners with academic, industry, and government partners to rapidly deploy new technologies to facilitate patient care and improve the lives of people suffering from neurological and behavioral health conditions.
GIVING OPPORTUNITIES
WVU Rockefeller Neuroscience Institute is forging the frontiers in neuroscience research…because of you.
Your generosity provides the needed momentum for the WVU Rockefeller Neuroscience Institute to leverage cutting-edge technologies and build strategic alliances to overcome the barriers to resolving some of the most persistent public health challenges, ranging from addiction to Alzheimer’s disease.